MD Diagnostics Ltd, UK design and manufacture a range of handheld Breath Carbon Monoxide monitors, Hydrogen breath monitors and Respiratory Pressure Meters including a wide range of consumables.
MD Diagnostics Ltd, UK design and manufacture a range of handheld Breath Carbon Monoxide monitors, Hydrogen breath monitors and Respiratory Pressure Meters including a wide range of consumables.
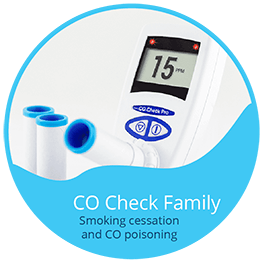
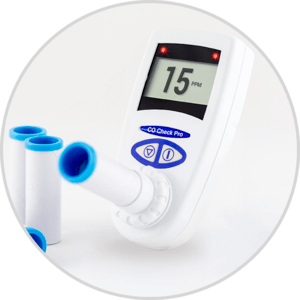
The CO Check breath monitors can be used for all Smoking Cessation purposes. A full range of breath Carbon Monoxide monitors include the CO Check Baby specifically designed for midwives to assess smoking in pregnancy by measuring the foetal carboxyhaemoglobin level. The range also includes the low cost CO Check + and full featured CO Check Pro with clear “traffic light indicators” of smoking status.
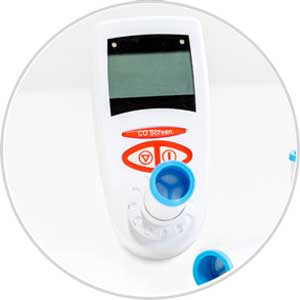
For Emergency Services and First Emergency Responders the CO Screen has been developed to instantly monitor potential CO poisoning using a quick and simple breath test.
For Dental Professionals we have the Dental CO. Dental professionals have a unique opportunity to address smoking with patients in a manner that will make a difference.
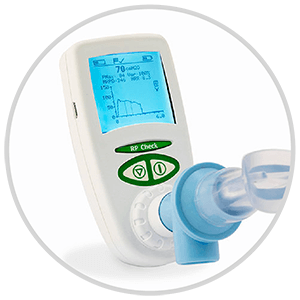
The RP Check is a handheld respiratory pressure meter measuring mouth and nasal pressures (MIP, MEP and SNIP). It is the only handheld device that can show pressure traces of each test on its large backlit display along with reproducibility criteria as per ATS/ERS.
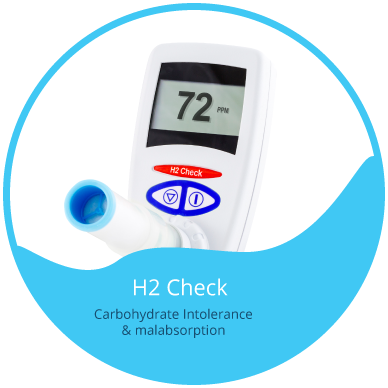
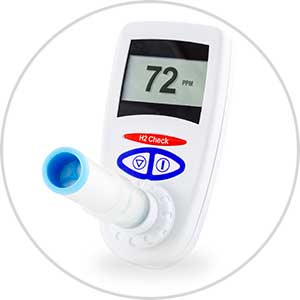
The H2 Check is a low cost simple to use Hydrogen breath test for Sugar or Carbohydrate Malabsorption / Intolerance. The H2 Check is suitable for all age groups of patients.
![]()